Physics for Everyone: Radiation
One of the most abused and least properly understood words in physics is “radiation.” When we hear that word, we immediately conjure in our minds images of some invisible bogie-man that’s going to melt our flesh and forever render us dead, sterile, or some horrible mutated version of our former selves. The fact of the matter is, there are many kinds of radiation, the majority of which are completely harmless (more or less), and in many of which we are constantly bathed. And while some types of radiation can be quite deadly should we become exposed to them in sufficient dosages, other varieties we couldn’t even exist without.
So what exactly is radiation? Simply put, it is that which is emitted by something. It can be a particle or a form of energy. The source, depending on what is causing it to do so, can radiate heat, light, subatomic particles, or in some cases, even ionized atoms (essentially just a nucleus – the atom with the electrons stripped off, or missing). So let’s address these one at a time and determine just which ones can hurt us, which can’t, and which are vital. And how each of them “spreads,” if they do at all.
Thermal Radiation
We’ll start with thermal radiation, or as it is thought of in common parlance, heat. Really, heat is just the flow of energy form one substance to another, usually through means of kinetic transfer- basically, the particles bump into each other while zipping about all willy nilly (which individual particles are wont to do). When the particles with more energy (either because they’re moving faster, or they’re more massive, or both), slam into the less energetic ones, they give up some of that energy to them. This will cause the energy to flow from the more energetic portions of the substance to the less energetic portions – heat.
Now, a “hot” atom (one that’s fairly energetic) can give up its energy in one other mean than just slamming into something else – if it has enough energy, its electrons can become excited. This will make them move into higher “orbits.” These electrons, while excitable, don’t want to stay in these higher orbits – they really want to be closer to the atom to which they’re bound. But to do this, the atom has to give up some energy, and it does so by emitting a photon as the electron falls from one orbit to a lower one, becoming less agitated. When it was merely kinetic energy exciting the atom, the drop is usually a fairly low-energy drop (as it goes), and the photon emitted will be in the infrared wavelength – what we tend to think of as thermal radiation.
As we all know, heat is generally not too bad. It keeps us warm on winter days. But, if it gets too much, it can hurt us (as anyone who’s been burned knows). As it goes, thermal radiation is fairly benign.
Electromagnetic Radiation
This is opening a can of worms. There are TONS of ways something can emit electromagnetic radiation, because that’s just the bigger fancier wording for “light.” Your flashlight emits it, your television, your computer, the sun, fire, fireflies, some fish, and fungus. All sorts of things emit light in a variety of ways. Obviously most of them are benign and really aren’t going to hurt us at all.
Just as with infrared radiation, some light is emitted when an electron falls from a higher energy level, or orbit around an atom, to a lower one. Just how large a drop that is determines the wavelength, and thereby energy, of the photon. The bigger the drop, the more energetic the photon. Most processes that emit visible light are from a lower energy process that causes this – it can be from heating up a metal wire by sending an electric current through it, such as in an incandescent light bulb, or a photo-luminescent process wherein a material absorbs photons and reemits them slowly, such as in glow-in-the-dark decals or toys. Bioluminescence is a different process wherein the animal or plant contains a chemical reaction that supplies the energy to emit light.
Some more energy electromagnetic radiation can come from much more violent process, however. This is where we start getting into dangerous stuff. Nuclear reactions, both fission and fusion, will general emit light –here it is not from electrons dropping orbits, but rather from them being stripped from the atom in entirety. Nuclear reactions can, and do, emit all wavelengths of light, from harmless radio-waves (whose wavelengths are so long they can’t really interact with are bodies), to the scary and highly energy gamma waves. But, to be honest, if you’re experiencing something as energetic as gamma radiation, it’s the least of your worries – you’re being expose to much more violent forces that will have more immediate concerns.
Nuclear Radiation
Now we’re going to get into the real bogey man.
There are really 4 types of nuclear radiation that we are generally concerned about – Alpha, Beta, Gamma, and Neutron. We’ve already gone over gamma radiation as a wavelength of light. Of the 4, it is the most penetrating, and definitely has the longest range - we can detect gamma radiation from highly energetic astronomical events from billions of light years away, but the others have ranges measured in feet or inches, or less.
Alpha radiation is a helium nucleus with two protons and two neutrons emitted either by a fission reaction or by the nuclear decay of a radioactive substance such as radon, radium, polonium, uranium, or thorium. It doesn’t have a very long range – alpha particles are usually absorbed after moving through only a few centimeters of air, or an even smaller distance of more solid material. Because of this short range, the only danger alpha radiation can pose to us is if we ingest an alpha emitter – which is highly unlikely unless you do so on purpose. And then, I have to ask where you got it. They’re not exactly common. So don’t worry about these guys.
Beta radiation has a bit longer range, though still very short - only a few millimeters of aluminum can stop it, and it won’t make it very far in air, either. This form of radiation is an electron or positron (the anti-electron, as it were), usually emitted when a nucleus that has too many neutrons solves its problem by having one of those protons “split” into a proton, an electron, and another particle called an antineutrino (don’t worry about the antineutrino – it, too, can be considered another type of radiation, but they, and their counterpart the neutrino, interact with matter so infrequently, that successful detection events in giant neutrino detectors are measured in single digits per year). The electron goes zipping off at an incredibly high rate speed, but is quickly snagged by another atom in whatever material it happens to encounter.
Neutron radiation is the one most people worry about without realizing it – and they worry about it WAY too much. It’s the one people are scared about spreading from Fukushima to California (and don’t worry, it won’t, and I’m about to tell you why). This form of radiation is exactly what it sounds like – a neutron that’s been ejected from a nucleus either by nuclear decay, or by fission where the nucleus was split by some external source, possible even another neutron. And this is why people worry about radiation spreading – neutron radiation actually can. It’s the only form of radiation that can make other objects radioactive, and it does so by a process called neutron activation.
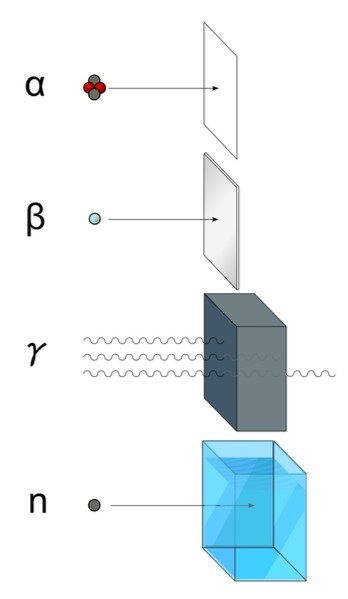
What the heck is that? Neutron activation is a process wherein a free-moving neutron is captured by an atomic nucleus. Now, most nuclei are already stable and have plenty of neutrons already. They don’t need another one. For some of them, introducing that new neutron to the neighborhood can cause plenty of drama in the nucleus, starting a decay process which may emit a beta particle, a proton, or even another neutron. Which can then be captured by another nucleus, and the whole process can possibly start all over again. This is usually what we’re talking about when we talk about chain reactions in a nuclear bomb, or in a reactor.
That sounds like scary stuff? Won’t the neutron activation process just keep going in one long chain reaction?
Thankfully, no. Not all elements are prone to neutron activation. For some, adding a neutron to their nucleus makes them MORE stable. So, essentially, what’ll end up happening is those neutrons being emitted will eventually run into elements that like them and want to keep them around, stopping any chance at a chain reaction (and that’s exactly why the Fukushima neutrons won’t make it to California – there’s a LOT of ocean in between Japan and North America).
Others
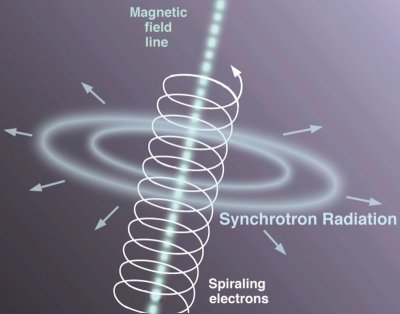
There are other types of radiation, most of them even more exotic than nuclear radiations. There’s Synchrotron Radiation, which is light emitted by electrons that are moving VERY close to the speed of light while being forced to move in a curved path by a magnetic field (generally harmless, and actually considered a nuisance in particle accelerator experiments, though detecting it in astronomical objects is useful as it tells us something about them). Cherenkov Radiation is really nifty because it happens when an electron actually manages to go faster than a photon can in certain media (but wait, didn’t I say that can’t happen in a previous article?!) – which can occur in places like nuclear reactors, where beta radiation is released, and the electron actually makes it through the water a bit before reacting with it. Hawking Radiation occurs at the event horizons of black holes – where particle pairs are created out of the black hole’s gravitic energy – the unlucky one to be on the wrong side of the event horizon gets sucked in while its partner goes zipping off into the cosmos at very near the speed of light. Cosmic radiation would be very dangerous if we didn’t have the Earth’s magnetic field and atmosphere to protect us from it – most of it is protons, while about 10% of it is alpha particles.

How do we know what radiation is dangerous?
Ultimately it boils down to whether or not the radiation is ionizing, and just how far it penetrates. All of the nuclear radiations are ionizing, but only two of them have any penetration or range. Neutron and Gamma radiation are going to be the biggest problems from radioactive materials, as neutron radiation can travel upwards of a couple kilometers, and gamma radiation an indefinite range unless it runs into a dense enough material. Higher wavelengths of light can be ionizing.
What makes ionizing radiations dangerous is the fact that they strip electrons away from atoms, creating a net positive charge. This can be harmful to organisms as it can affect our DNA, or cause adverse chemical reactions in our bodies.
Non-ionizing radiations don’t wreak havoc with our DNA or body chemistry quite the way ionizing radiations do, but in sufficient intensities, can still cause harm – just try putting your hand on a stove element. Or don’t, actually – we all know that ends badly.
Radiation comes in many forms, and not all of it bad. Some of it’s quite beneficial – and we wouldn’t be here without it. So, really, stop worrying so much when a scientist mentions the word radiation, and stop and listen to what they’re talking about – it might not be one of the scary types.
|



